ND-FS
NeoDrape Fenestrated
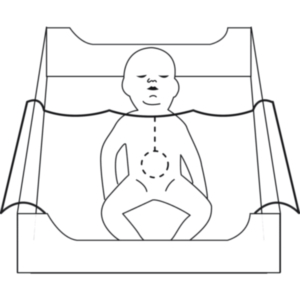
The NEOMED* Fenestrated Sterile Drape is intended as a sterile protective patient covering to isolate a procedure site from contamination. The NEOMED* Sterile Drape is intended to be used as an occlusive thermal wrap.
Resources
| Name | Value |
|---|---|
| Primary Device Identifier (GTIN) | 00350770003375 |
| Product Name | NeoDrape Fenestrated |
| Prescription Use (Rx) | True |
| Product Type | Procedure Drapes |
| Requires Sterilization Prior to Use | False |
| Sterile | True |
| Sterilization Method | Ethylene Oxide |
| Name | Value |
|---|---|
| Selling Package Details | 1 CS = 40 Units |
| Selling Package Volume | 18.120 CDM |
| Selling Package Length | 22.860 CM |
| Selling Package Width | 29.720 CM |
| Selling Package Height | 26.670 CM |
| Selling Package Weight | 1.588 KG |
Provide your information below so we can put you in touch with the correct Avanos Representative who will help you with ordering information:

ND-FS
NeoDrape Fenestrated
Product Overview
The NEOMED* Fenestrated Sterile Drape is intended as a sterile protective patient covering to isolate a procedure site from contamination. The NEOMED* Sterile Drape is intended to be used as an occlusive thermal wrap.Product Features
Specifications
| Name | Value |
|---|---|
| Primary Device Identifier (GTIN) | 00350770003375 |
| Product Name | NeoDrape Fenestrated |
| Prescription Use (Rx) | True |
| Product Type | Procedure Drapes |
| Requires Sterilization Prior to Use | False |
| Sterile | True |
| Sterilization Method | Ethylene Oxide |
Packaging Specifications
| Name | Value |
|---|---|
| Selling Package Details | 1 CS = 40 Units |
| Selling Package Volume | 18.120 CDM |
| Selling Package Length | 22.860 CM |
| Selling Package Width | 29.720 CM |
| Selling Package Height | 26.670 CM |
| Selling Package Weight | 1.588 KG |
Customer Service and Ordering Information
For customer service or ordering information, please contact us at 1 844 4Avanos
The dimensions and properties listed can vary within pre-established specifications. This page was created using the most recent information. In the interest of continuous improvement, the characteristics of the product may change without prior notice.
The dimensions and properties listed can vary within pre-established specifications. This page was created using the most recent information. In the interest of continuous improvement, the characteristics of the product may change without prior notice.
















 Cooled Radiofrequency Ablation (RFA)
Cooled Radiofrequency Ablation (RFA) Tined Radiofrequency Ablation (RFA)
Tined Radiofrequency Ablation (RFA) Conventional Radiofrequency Ablation (RFA)
Conventional Radiofrequency Ablation (RFA) RFA Equipment & Accessories
RFA Equipment & Accessories Needles, Kits, and Trays
Needles, Kits, and Trays Elastomeric Pain Pumps and Kits
Elastomeric Pain Pumps and Kits Needles, Kits, and Trays (Incisional and PNB)
Needles, Kits, and Trays (Incisional and PNB) Cryo Compression Control Unit Systems
Cryo Compression Control Unit Systems Cryo Compression Sleeves and Wraps
Cryo Compression Sleeves and Wraps Feeding Tube Placement Device
Feeding Tube Placement Device Feeding Tube Retention Systems
Feeding Tube Retention Systems Declogging and Decompression
Declogging and Decompression Feeding Tubes and Accessories
Feeding Tubes and Accessories Introducer Kits and Accessories
Introducer Kits and Accessories Feeding Tube Extension Sets and Adapters
Feeding Tube Extension Sets and Adapters Enteral Medication Preparation & Delivery
Enteral Medication Preparation & Delivery Feeding Accessories
Feeding Accessories Feeding Pumps
Feeding Pumps Feeding Syringes
Feeding Syringes Feeding Tubes and Extension Sets
Feeding Tubes and Extension Sets Lumbar Puncture Trays and Needles
Lumbar Puncture Trays and Needles Oral Care/Colostrum Kits and Components
Oral Care/Colostrum Kits and Components Urinary Catheters & Kits
Urinary Catheters & Kits