7150-14
MIC* Percutaneous Endoscopic Gastrostomy PEG Kit – 14 Fr – PUSH OTW
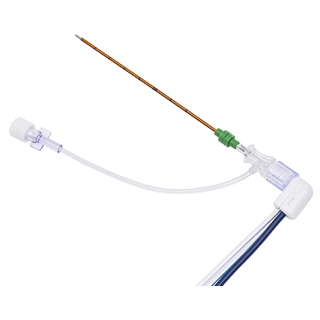
Gastrostomy tube feeding may be indicated for patients needing long-term enteral support or hydration secondary to a primary condition relating to the head and/or neck. These conditions include stroke; cancer; head and neck tumors, injuries, or trauma; and neurological disorders resulting in a chewing or swallowing abnormality. This device (sold in a kit) is intended as an initial placement device. The device is placed by one of two techniques, the PULL technique and the over-the-guidewire technique (PUSH technique).
Resources
| Sterile Kit Contents: |
| 1 PEG Tube |
| 1 Retrieval Snare |
| 1 Feeding Adapter |
| 1 Lidocaine, 1% HCI, USP Vial 5 ml |
| 1 Filter Needle, 19 gauge x 1-1/2 in. 5 micron |
| 1 Injection Needle, 25 gauge x 1.5 in. |
| 1 External Retention Bolster |
| 1 Chloraprep® Swabstick Packet |
| 1 Fenestrated Drape |
| 1 Syringe, 6 ml |
| 1 Scalpel, #11 Blade |
| 1 Introducer Needle, with Cannula 14 gauge |
| 1 Guidewire |
| 1 Stainless Steel Scissors |
| 1 Stainless Steel Hemostat |
| 1 Tubing Clamp |
| 1 Water Soluble Lubricant |
| 4 Gauze Sponges, 4 in. x 4 in. |
| 2 Slit Gauze Sponges, 2 in. x 2 in. |
| 1 Lidocaine Label |
| Non-Sterile Kit Contents: 1 Povidone-Iodine Swabstick Packet |
| Name | Value |
|---|---|
| Primary Device Identifier (GTIN) | 00350770006130 |
| ENFit* Connector | No |
| Medication Port | Yes |
| Prescription Use (Rx) | True |
| Product Name | MIC* Percutaneous Endoscopic Gastrostomy PEG Kit – 14 Fr – PUSH OTW |
| Product Type | Feeding Tubes |
| Standard or Safety Kit | Standard |
| Sterile | True |
| Sterilization Method | Ethylene Oxide |
| Tube Outer Diameter (Fr) | 14 |
| Tubing Clamp | 2 |
| Name | Value |
|---|---|
| Selling Package Details | 1 CS = 2 Units |
| Selling Package Volume | 10.540 CDM |
| Selling Package Length | 33.782 CM |
| Selling Package Width | 23.622 CM |
| Selling Package Height | 13.208 CM |
| Selling Package Weight | 0.998 KG |
Provide your information below so we can put you in touch with the correct Avanos Representative who will help you with ordering information:


7150-14
MIC* Percutaneous Endoscopic Gastrostomy PEG Kit – 14 Fr – PUSH OTW
Product Overview
Gastrostomy tube feeding may be indicated for patients needing long-term enteral support or hydration secondary to a primary condition relating to the head and/or neck. These conditions include stroke; cancer; head and neck tumors, injuries, or trauma; and neurological disorders resulting in a chewing or swallowing abnormality. This device (sold in a kit) is intended as an initial placement device. The device is placed by one of two techniques, the PULL technique and the over-the-guidewire technique (PUSH technique).Product Features
Kit Contents
| Sterile Kit Contents: |
| 1 PEG Tube |
| 1 Retrieval Snare |
| 1 Feeding Adapter |
| 1 Lidocaine, 1% HCI, USP Vial 5 ml |
| 1 Filter Needle, 19 gauge x 1-1/2 in. 5 micron |
| 1 Injection Needle, 25 gauge x 1.5 in. |
| 1 External Retention Bolster |
| 1 Chloraprep® Swabstick Packet |
| 1 Fenestrated Drape |
| 1 Syringe, 6 ml |
| 1 Scalpel, #11 Blade |
| 1 Introducer Needle, with Cannula 14 gauge |
| 1 Guidewire |
| 1 Stainless Steel Scissors |
| 1 Stainless Steel Hemostat |
| 1 Tubing Clamp |
| 1 Water Soluble Lubricant |
| 4 Gauze Sponges, 4 in. x 4 in. |
| 2 Slit Gauze Sponges, 2 in. x 2 in. |
| 1 Lidocaine Label |
| Non-Sterile Kit Contents: 1 Povidone-Iodine Swabstick Packet |
Specifications
| Name | Value |
|---|---|
| Primary Device Identifier (GTIN) | 00350770006130 |
| ENFit* Connector | No |
| Medication Port | Yes |
| Prescription Use (Rx) | True |
| Product Name | MIC* Percutaneous Endoscopic Gastrostomy PEG Kit – 14 Fr – PUSH OTW |
| Product Type | Feeding Tubes |
| Standard or Safety Kit | Standard |
| Sterile | True |
| Sterilization Method | Ethylene Oxide |
| Tube Outer Diameter (Fr) | 14 |
| Tubing Clamp | 2 |
Packaging Specifications
| Name | Value |
|---|---|
| Selling Package Details | 1 CS = 2 Units |
| Selling Package Volume | 10.540 CDM |
| Selling Package Length | 33.782 CM |
| Selling Package Width | 23.622 CM |
| Selling Package Height | 13.208 CM |
| Selling Package Weight | 0.998 KG |
Additional Product Images


Customer Service and Ordering Information
For customer service or ordering information, please contact us at 1 844 4Avanos
The dimensions and properties listed can vary within pre-established specifications. This page was created using the most recent information. In the interest of continuous improvement, the characteristics of the product may change without prior notice.
The dimensions and properties listed can vary within pre-established specifications. This page was created using the most recent information. In the interest of continuous improvement, the characteristics of the product may change without prior notice.
















 Cooled Radiofrequency Ablation (RFA)
Cooled Radiofrequency Ablation (RFA) Tined Radiofrequency Ablation (RFA)
Tined Radiofrequency Ablation (RFA) Conventional Radiofrequency Ablation (RFA)
Conventional Radiofrequency Ablation (RFA) RFA Equipment & Accessories
RFA Equipment & Accessories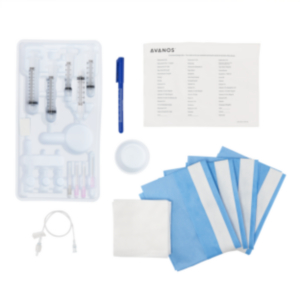 Needles, Kits, and Trays
Needles, Kits, and Trays Elastomeric Pain Pumps and Kits
Elastomeric Pain Pumps and Kits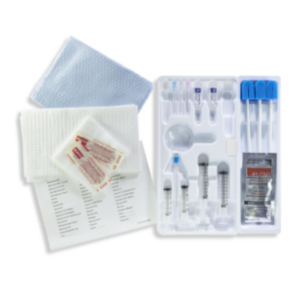 Needles, Kits, and Trays (Incisional and PNB)
Needles, Kits, and Trays (Incisional and PNB) Cryo Compression Control Unit Systems
Cryo Compression Control Unit Systems Cryo Compression Sleeves and Wraps
Cryo Compression Sleeves and Wraps Feeding Tube Placement Device
Feeding Tube Placement Device Feeding Tube Retention Systems
Feeding Tube Retention Systems Declogging and Decompression
Declogging and Decompression Feeding Tubes and Accessories
Feeding Tubes and Accessories Introducer Kits and Accessories
Introducer Kits and Accessories Feeding Tube Extension Sets and Adapters
Feeding Tube Extension Sets and Adapters Enteral Medication Preparation & Delivery
Enteral Medication Preparation & Delivery Feeding Accessories
Feeding Accessories Feeding Pumps
Feeding Pumps Feeding Syringes
Feeding Syringes Feeding Tubes and Extension Sets
Feeding Tubes and Extension Sets Lumbar Puncture Trays and Needles
Lumbar Puncture Trays and Needles Oral Care/Colostrum Kits and Components
Oral Care/Colostrum Kits and Components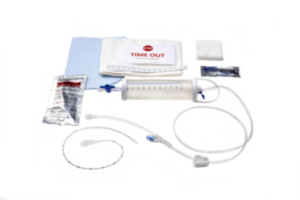 Urinary Catheters & Kits
Urinary Catheters & Kits